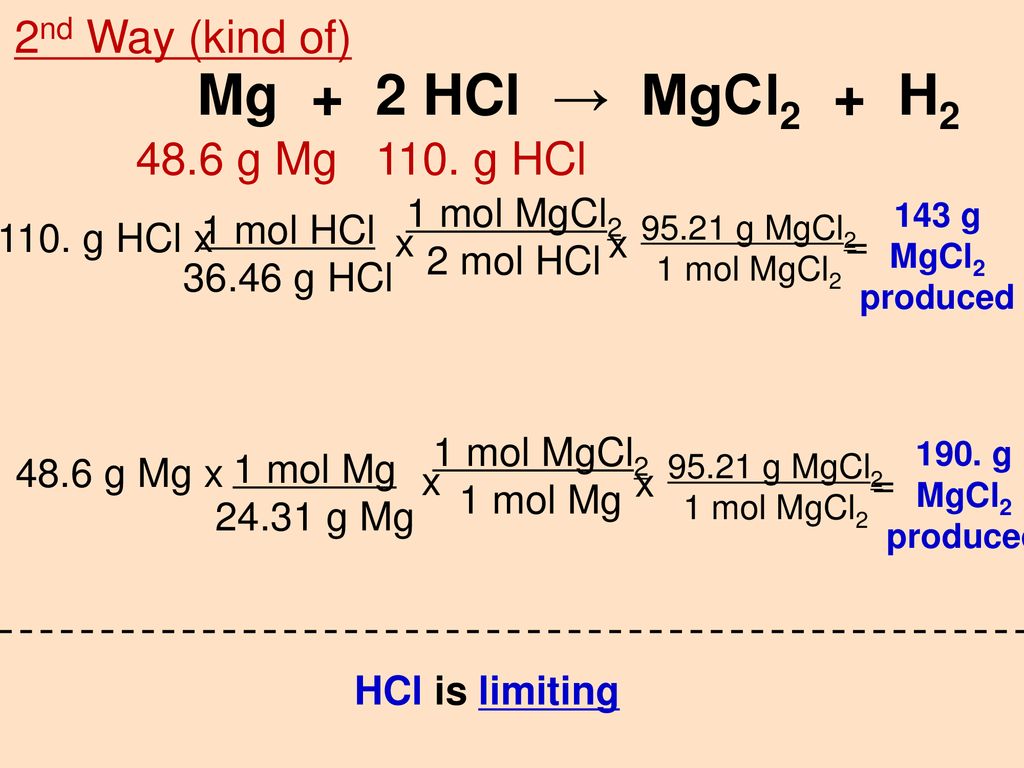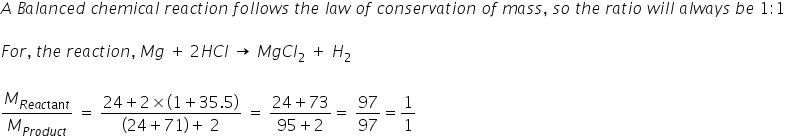
calculate the ratios of the masses of the reactants and products in the following reaction mg 2hcl mgcl2 h2 - Chemistry - TopperLearning.com | c3tctzii

SOLVED:Given the reaction: Mg(s) + 2 HCl(aq) â†' MgCl2(aq) + H2(g) Which species is reduced? MgCl2 H2 HCl Mg

Balance the following equations.(i) KMnO2 + HCl → KCl + MnCl2 + H2 O + Cl2 (ii) NH3 + O3 → NO + H2 O

SOLVED:Use the balanced chemical equation to determine how many grams of MgCl2 are being produced if you begin with 5.55 g of Mg and the reaction runs with a 60% yield. Mg +

Balance the following equations.(i) KMnO2 + HCl → KCl + MnCl2 + H2 O + Cl2 (ii) NH3 + O3 → NO + H2 O

According to the chemical equation, mg(s) + 2hcl mgcl2(aq) + h2(g) how many moles of magnesium are - Brainly.in


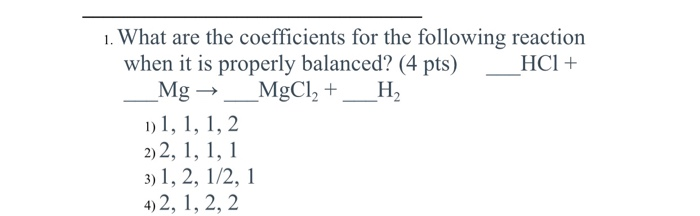



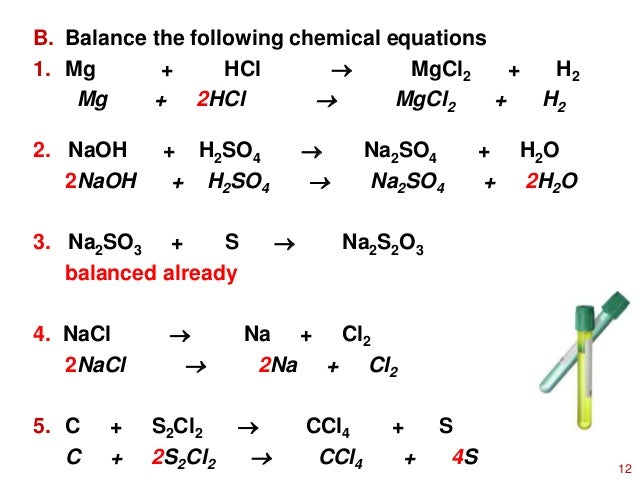




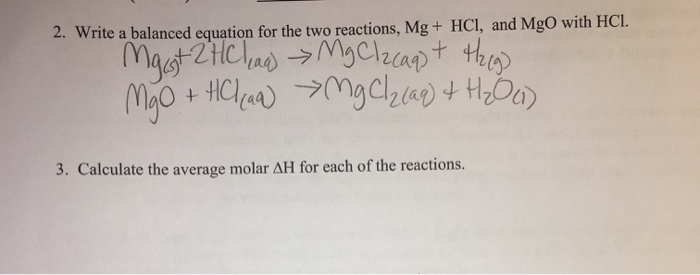
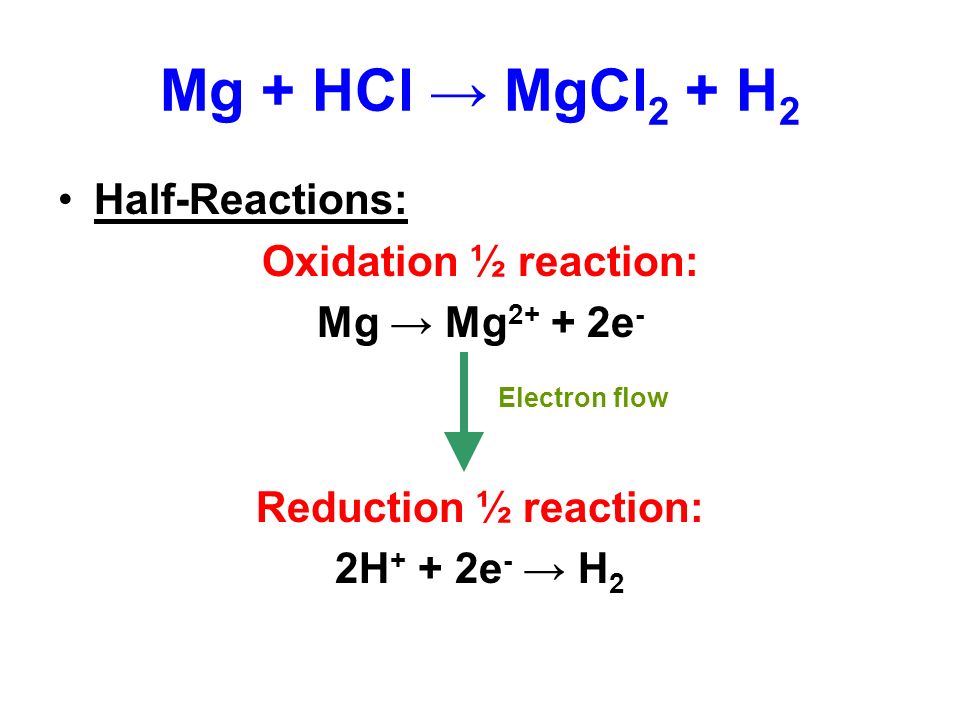
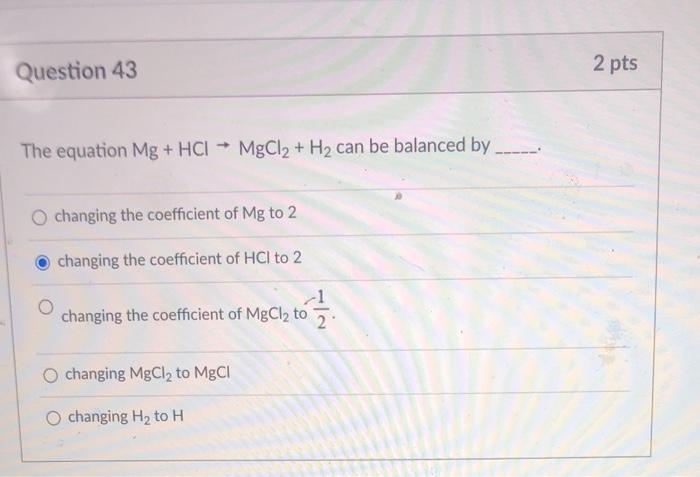
![PPT - Do Now – April [2(10) + 3], 2009 PowerPoint Presentation, free download - ID:3888096 PPT - Do Now – April [2(10) + 3], 2009 PowerPoint Presentation, free download - ID:3888096](https://image2.slideserve.com/3888096/slide24-l.jpg)